12 Recent Discoveries in Alzheimer's Research That Could Change Treatment
The landscape of Alzheimer's disease research has undergone a revolutionary transformation in recent years, with groundbreaking discoveries challenging long-held assumptions about the nature, progression, and potential treatment of this devastating neurodegenerative condition. For decades, researchers have grappled with the complex mechanisms underlying Alzheimer's disease, primarily focusing on the accumulation of amyloid plaques and tau tangles in the brain. However, recent scientific breakthroughs have unveiled a multifaceted picture of the disease that extends far beyond these traditional hallmarks, encompassing everything from the role of the immune system and sleep patterns to the influence of gut bacteria and novel therapeutic targets. These discoveries are not merely academic curiosities; they represent potential paradigm shifts that could fundamentally alter how we diagnose, prevent, and treat Alzheimer's disease. The following twelve recent discoveries represent some of the most promising and transformative findings in the field, each offering unique insights that collectively paint a more comprehensive and hopeful picture of our fight against this debilitating condition that affects millions of people worldwide.
1. The Glymphatic System - Brain's Hidden Cleaning Network
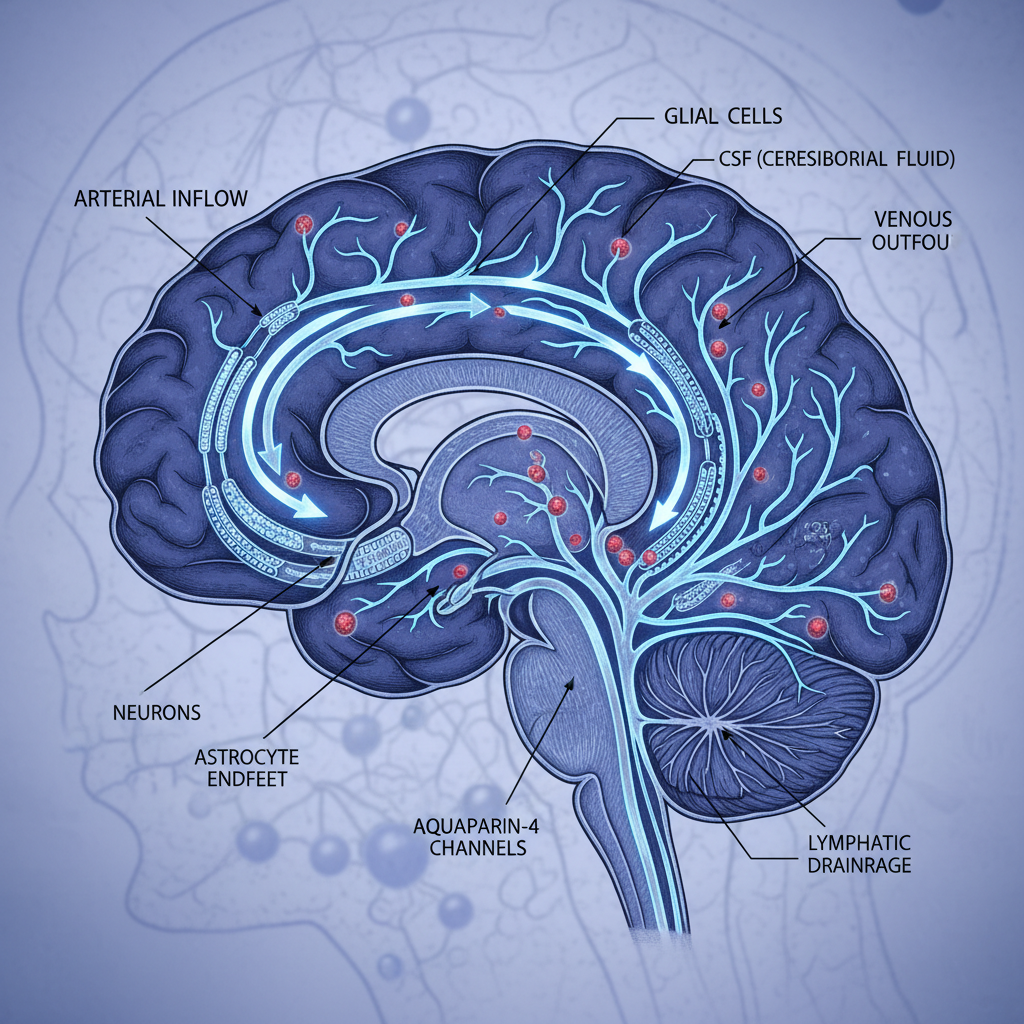
One of the most significant recent discoveries in Alzheimer's research has been the identification and characterization of the glymphatic system, the brain's previously unknown waste clearance mechanism that operates primarily during sleep. This intricate network of fluid channels, discovered through advanced neuroimaging techniques, functions as the brain's equivalent of the lymphatic system, efficiently removing metabolic waste products including amyloid-beta proteins and other toxic substances that accumulate during waking hours. Researchers have found that the glymphatic system becomes significantly more active during deep sleep stages, when brain cells shrink by up to 60%, creating expanded spaces between neurons that allow cerebrospinal fluid to flow more freely and flush out harmful proteins. This discovery has profound implications for Alzheimer's prevention and treatment, as it suggests that sleep quality and duration may be far more critical to brain health than previously understood. Studies have shown that individuals with chronic sleep disorders or poor sleep quality have significantly higher levels of amyloid-beta accumulation in their brains, potentially accelerating the onset and progression of Alzheimer's disease. This finding has led to the development of new therapeutic approaches focused on enhancing glymphatic function through sleep optimization, positioning strategies, and even pharmacological interventions that could help maintain this crucial brain cleaning system throughout aging.
2. Neuroinflammation and Microglial Activation

The role of neuroinflammation in Alzheimer's disease has emerged as a critical area of research, fundamentally shifting our understanding from viewing inflammation as merely a consequence of the disease to recognizing it as a potential driving force in its development and progression. Microglia, the brain's resident immune cells, have been found to play a dual role in Alzheimer's pathology, initially serving as protective agents that attempt to clear amyloid plaques and damaged neurons, but eventually becoming chronically activated and contributing to widespread brain inflammation. Recent studies using advanced molecular imaging techniques have revealed that microglial activation occurs years before clinical symptoms appear, suggesting that neuroinflammation may be one of the earliest detectable changes in the Alzheimer's disease process. This chronic inflammatory state creates a vicious cycle where activated microglia release pro-inflammatory cytokines and reactive oxygen species that damage healthy neurons, while simultaneously losing their ability to effectively clear amyloid-beta and tau proteins. The discovery of specific genetic variants in microglial genes, such as TREM2 and CD33, that significantly alter Alzheimer's risk has further highlighted the importance of immune dysfunction in disease development. These findings have opened new avenues for therapeutic intervention, with researchers developing anti-inflammatory drugs, microglial modulators, and immunotherapies designed to restore the brain's immune balance and potentially slow or halt disease progression.
3. The Gut-Brain Axis and Microbiome Connections

Revolutionary research has unveiled the profound connection between the gut microbiome and Alzheimer's disease, establishing what scientists now call the "gut-brain axis" as a critical pathway in neurodegeneration. The human gut contains trillions of microorganisms that produce various metabolites, neurotransmitters, and inflammatory compounds that can directly influence brain function through multiple pathways including the vagus nerve, immune system modulation, and the production of short-chain fatty acids that can cross the blood-brain barrier. Recent studies have identified specific bacterial strains and microbiome compositions that are consistently altered in individuals with Alzheimer's disease, with reduced microbial diversity and increased populations of pro-inflammatory bacteria being common findings. Particularly significant is the discovery that certain gut bacteria can produce amyloid proteins similar to those found in Alzheimer's plaques, potentially seeding the formation of pathological protein aggregates in the brain through a process called molecular mimicry. Additionally, the gut microbiome plays a crucial role in producing and metabolizing neurotransmitters such as serotonin, dopamine, and GABA, which are essential for cognitive function and mood regulation. This research has led to the development of novel therapeutic approaches including targeted probiotic treatments, fecal microbiota transplantation, and dietary interventions designed to restore healthy gut bacteria populations. Clinical trials are currently investigating whether modifying the gut microbiome through these interventions can slow cognitive decline and improve outcomes in Alzheimer's patients.
4. Tau Protein Spreading and Prion-Like Propagation

The discovery that tau protein, one of the key pathological hallmarks of Alzheimer's disease, can spread throughout the brain in a prion-like manner has revolutionized our understanding of disease progression and opened new therapeutic possibilities. Unlike the random accumulation previously assumed, researchers have found that misfolded tau proteins can act as templates that induce normal tau proteins to adopt abnormal conformations, creating a cascade of protein misfolding that spreads from cell to cell along neural networks. This propagation follows predictable patterns that correlate closely with the clinical stages of Alzheimer's disease, beginning in the entorhinal cortex and hippocampus before spreading to other brain regions involved in memory and cognition. Advanced neuroimaging techniques using tau-specific PET tracers have allowed scientists to visualize this spreading process in living patients, revealing that tau pathology correlates more strongly with cognitive symptoms than amyloid plaques. The prion-like nature of tau spreading suggests that different strains of misfolded tau may exist, potentially explaining the heterogeneity observed in Alzheimer's disease presentation and progression rates among different patients. This discovery has significant implications for treatment timing and strategy, as it suggests that interventions targeting tau propagation may be most effective in the early stages of disease before widespread spreading has occurred. Researchers are now developing therapies designed to block tau transmission between cells, including antibodies that can neutralize extracellular tau, small molecules that prevent tau aggregation, and approaches to enhance cellular mechanisms for clearing misfolded proteins.
5. Blood-Brain Barrier Dysfunction

Recent research has identified blood-brain barrier dysfunction as a critical early event in Alzheimer's disease development, fundamentally changing our understanding of how the disease begins and progresses. The blood-brain barrier, a highly selective semipermeable membrane that protects the brain from harmful substances while allowing essential nutrients to pass through, becomes increasingly compromised with age and disease. Advanced imaging techniques have revealed that blood-brain barrier breakdown occurs years before clinical symptoms appear and correlates strongly with cognitive decline and brain atrophy. This dysfunction allows toxic substances from the blood to enter the brain while preventing the efficient removal of waste products, creating an environment conducive to neurodegeneration. Researchers have discovered that specific proteins involved in maintaining blood-brain barrier integrity, such as claudin-5 and occludin, are significantly reduced in Alzheimer's patients, while inflammatory mediators that increase vascular permeability are elevated. The breakdown of this protective barrier also allows peripheral immune cells to infiltrate the brain, contributing to neuroinflammation and tissue damage. Additionally, blood-brain barrier dysfunction impairs the delivery of potential therapeutic agents to the brain, which may partially explain the failure of many Alzheimer's treatments in clinical trials. This discovery has led to the development of new therapeutic strategies focused on restoring blood-brain barrier function through targeted drug delivery systems, vascular protective agents, and interventions designed to strengthen the cellular junctions that maintain barrier integrity.
6. Cellular Senescence and Aging Mechanisms

The identification of cellular senescence as a key driver of Alzheimer's disease has provided new insights into the fundamental aging processes that contribute to neurodegeneration. Senescent cells are aged cells that have stopped dividing but remain metabolically active, secreting a complex mixture of inflammatory factors, proteases, and growth factors known as the senescence-associated secretory phenotype (SASP). These senescent cells accumulate in the brain with age and have been found in significantly higher numbers in individuals with Alzheimer's disease, particularly in regions most affected by the disease such as the hippocampus and cortex. The SASP factors released by senescent cells create a chronic inflammatory environment that damages healthy neurons, promotes tau pathology, and impairs the function of other brain cells including microglia and astrocytes. Recent studies have shown that senescent cells can also disrupt normal brain metabolism, reduce neuroplasticity, and interfere with the brain's ability to form new neural connections. Perhaps most importantly, researchers have discovered that selectively eliminating senescent cells using drugs called senolytics can significantly improve cognitive function and reduce pathological changes in animal models of Alzheimer's disease. This has led to clinical trials investigating whether senolytic therapies, such as dasatinib and quercetin combinations, can slow cognitive decline in humans. The senescence pathway also offers new biomarkers for early disease detection and monitoring treatment response, as senescence markers in blood and cerebrospinal fluid correlate with disease severity and progression.
7. Mitochondrial Dysfunction and Energy Metabolism

Groundbreaking research has revealed that mitochondrial dysfunction and altered brain energy metabolism play central roles in Alzheimer's disease pathogenesis, offering new therapeutic targets for intervention. Mitochondria, the cellular powerhouses responsible for producing ATP energy, become increasingly dysfunctional in Alzheimer's disease, leading to reduced energy production, increased oxidative stress, and impaired cellular maintenance processes. Advanced metabolic imaging studies have shown that brain glucose metabolism is significantly reduced in Alzheimer's patients, often years before clinical symptoms appear, suggesting that energy deficits may be among the earliest detectable changes in the disease process. Researchers have discovered that amyloid-beta and tau proteins directly interact with mitochondria, disrupting their structure and function, while mitochondrial dysfunction in turn promotes the production and aggregation of these pathological proteins, creating a destructive feedback loop. The brain's high energy demands make neurons particularly vulnerable to mitochondrial dysfunction, and specific brain regions with the highest metabolic requirements, such as the hippocampus, are often the first to be affected in Alzheimer's disease. Recent studies have also revealed that mitochondrial DNA mutations accumulate with age and are more prevalent in Alzheimer's patients, potentially contributing to the progressive nature of the disease. This understanding has led to the development of novel therapeutic approaches including mitochondrial-targeted antioxidants, metabolic enhancers that improve cellular energy production, and ketogenic interventions that provide alternative fuel sources for the brain when glucose metabolism is impaired.
8. Epigenetic Modifications and Gene Expression Changes

The discovery of widespread epigenetic alterations in Alzheimer's disease has opened an entirely new dimension of understanding about how environmental factors and aging processes influence disease development and progression. Epigenetic modifications, which alter gene expression without changing the underlying DNA sequence, have been found to be extensively dysregulated in Alzheimer's patients, affecting thousands of genes involved in neuronal function, inflammation, and protein homeostasis. Recent genome-wide studies have identified specific patterns of DNA methylation and histone modifications that correlate with disease severity and can predict cognitive decline years before clinical symptoms appear. Particularly significant is the discovery that genes involved in synaptic function and neuroplasticity show consistent epigenetic silencing in Alzheimer's disease, while genes promoting inflammation and cell death become epigenetically activated. Environmental factors such as diet, exercise, stress, and education have been found to influence these epigenetic patterns, potentially explaining why lifestyle factors can significantly impact Alzheimer's risk and progression. The reversible nature of epigenetic modifications makes them particularly attractive therapeutic targets, as interventions that restore normal epigenetic patterns could potentially reverse some aspects of disease pathology. Researchers are developing epigenetic therapies including DNA methyltransferase inhibitors, histone deacetylase inhibitors, and targeted approaches to reactivate silenced neuroprotective genes. Additionally, epigenetic biomarkers in blood and cerebrospinal fluid are being investigated as tools for early diagnosis and monitoring treatment response, offering the possibility of personalized medicine approaches based on individual epigenetic profiles.
9. Synaptic Dysfunction and Neural Network Disruption

Revolutionary advances in understanding synaptic dysfunction have revealed that the loss of connections between neurons, rather than neuronal death itself, may be the primary driver of cognitive symptoms in Alzheimer's disease. Sophisticated neuroimaging techniques and electrophysiological studies have shown that synaptic dysfunction occurs very early in the disease process, often preceding the formation of amyloid plaques and neurofibrillary tangles by years or even decades. Researchers have discovered that oligomeric forms of amyloid-beta, small soluble clusters of the protein, are particularly toxic to synapses, disrupting neurotransmitter release, impairing synaptic plasticity, and ultimately leading to synapse loss. This synaptic damage preferentially affects specific neural networks, particularly the default mode network involved in memory consolidation and self-referential thinking, explaining why memory problems are often the first clinical manifestation of Alzheimer's disease. Advanced connectivity analyses have revealed that the brain's network organization becomes increasingly disrupted as the disease progresses, with reduced communication between distant brain regions and compensatory hyperactivation in some areas attempting to maintain function. The discovery that synaptic dysfunction is potentially reversible, even in the presence of some pathological protein accumulation, has significant therapeutic implications. New treatment approaches are being developed to enhance synaptic function through neurotransmitter modulation, synaptic protein stabilization, and interventions that promote the formation of new synaptic connections. Additionally, measures of synaptic function are being investigated as sensitive biomarkers for early disease detection and treatment monitoring.
10. Novel Biomarkers and Early Detection Methods

The development of novel biomarkers for Alzheimer's disease represents one of the most promising areas of recent research, with the potential to revolutionize early detection, diagnosis, and treatment monitoring. Traditional diagnostic approaches have relied heavily on cognitive testing and neuroimaging, which often detect the disease only after significant brain damage has already occurred. Recent breakthroughs have identified blood-based biomarkers that can detect Alzheimer's-related changes decades before clinical symptoms appear, including specific forms of amyloid-beta, tau proteins, and neurofilament light chain that reflect ongoing neurodegeneration. Advanced mass spectrometry techniques have enabled the detection of these proteins at extremely low concentrations in blood samples, making routine screening for Alzheimer's risk a realistic possibility. Particularly exciting is the development of phosphorylated tau biomarkers that can distinguish Alzheimer's disease from other forms of dementia with high accuracy, addressing a long-standing challenge in differential diagnosis. Researchers have also discovered novel biomarkers related to neuroinflammation, synaptic dysfunction, and metabolic changes that provide complementary information about different aspects of disease pathology. Digital biomarkers derived from smartphone apps, wearable devices, and computerized cognitive assessments are being developed to continuously monitor cognitive function and detect subtle changes that may indicate disease progression. The integration of multiple biomarker types into comprehensive panels is showing promise for predicting individual disease trajectories and treatment responses, enabling personalized medicine approaches. These advances are particularly important for clinical trial design, as they allow researchers to identify appropriate participants and monitor treatment effects more sensitively than traditional outcome measures.
11. Immunotherapy and Targeted Drug Delivery

The field of Alzheimer's immunotherapy has experienced a renaissance following recent successes and innovations in targeting disease-specific proteins and enhancing drug delivery to the brain. After years of disappointing results with amyloid-targeting antibodies, recent clinical trials have shown that newer generation immunotherapies, such as aducanumab and lecanemab, can significantly reduce amyloid plaques in the brain and, in some cases, slow cognitive decline. These successes have been attributed to improved antibody design, better patient selection using biomarker-guided approaches, and treatment initiation in earlier disease stages when interventions may be most effective. Researchers are now developing next-generation immunotherapies that target multiple pathological proteins simultaneously, including combination approaches that address both amyloid and tau pathology. Novel delivery methods are being employed to enhance antibody penetration into the brain, including focused ultrasound techniques that temporarily open the blood-brain barrier and nanoparticle formulations that can cross this protective barrier more efficiently. Passive immunization approaches using antibodies are being complemented by active immunization strategies that stimulate the patient's own immune system to produce antibodies against pathological proteins. Additionally, researchers are developing immunotherapies that target neuroinflammation, senescent cells, and other non-protein targets involved in Alzheimer's pathogenesis. The success of these approaches has reinvigorated interest in immunotherapy as a treatment modality and has led to increased investment in developing more effective and safer immunological interventions for Alzheimer's disease.
12. Precision Medicine and Personalized Treatment Approaches

The emergence of precision medicine in Alzheimer's research represents a fundamental shift from the traditional one-size-fits-all approach to treatment toward personalized interventions based on individual genetic, biological, and clinical characteristics. Recent advances in genomics, proteomics, and metabolomics have revealed that Alzheimer's disease is not a single condition but rather a heterogeneous group of disorders with different underlying mechanisms, progression patterns, and treatment responses. Genetic testing for risk variants such as APOE4, TREM2, and other susceptibility genes is being integrated with biomarker profiles to identify individuals who may benefit from specific therapeutic approaches. For example, APOE







